The need of ultrathin and highly conformal coatings on large-area microstructure substrates such as photovoltaic cells, flexible displays, and Li-ion battery (LIB) electrodes has driven the development of high-efficient ALD. Spatial ALD is a promising high-throughput technique capable of producing ultrathin films on large substrates, which can reach up to two orders of magnitude faster than temporal ALD. However, spatial ALD is a complex and strong-coupled process of fluid flow, heat and mass transfer, as well as chemical reactions. Compared to the flat substrate, the diffusion process in the microstructures takes longer, limiting the substrate velocity and the deposition rate. In this work, spatial ALD on moving microgroove substrates and porous LIB electrodes are quantitatively studied through the multiscale modelling method by coupling computational fluid dynamics (CFD) with chemical kinetics. Dynamic mesh method is implemented to simulate the ALD reaction with the in-line movement of the substrate. Results show that the vortices always exist in the micro-gap with microgroove substrates. Besides, the top of the microstructure on the first contact with the precursor is the highest, and the surface coverage at the corner on the other side of the microstructure is the lowest. For porous LIB electrodes with large specific surface area, the film coating depth is limited by both the low precursor diffusion rate and the amount of precursor supply. Increasing the carrier gas flow rate increases the film coating depth by increasing the precursor supply over the same time period. Besides, the gas byproduct of ALD remaining at the electrode bottom can be more effectively removed. Quantitative process optimizations of substrate velocity and precursor concentration for different electrode structures are also carried out. It is found that the electrode gradient design with higher porosity on the top is beneficial for obtaining deeper coating depth and higher precursor utilization compared to the inverse gradient design. The present multiscale modeling work provides a comprehensive understanding and important guidance for the industrial application of the spatial ALD process on microstructure substrates.

The Electrochemical Society (ECS) was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology, and allied subjects.
ISSN: 2151-2043
ECS Meeting Abstracts contain extended abstracts of the technical papers presented at ECS meetings and ECS-sponsored meetings. This publication offers a first look into the current research in the field. ECS Meeting Abstracts are freely available to all visitors to the ECS Digital Library.
Open all abstracts, in this tab
Zoushuang Li et al 2023 Meet. Abstr. MA2023-02 1473
Sviatlana Lamaka et al 2023 Meet. Abstr. MA2023-02 1136
Unravelling the mechanisms of magnesium corrosion is vital for establishing reliable research methodologies, developing new alloys and predicting magnesium degradation behaviour. Hydrogen evolution reaction is widely regarded as by far the main cathodic process during corrosion of magnesium. Hence, tracing the amount of released hydrogen was thought to be a reliable measure of magnesium degradation rate. Recently, experimental evidences obtained by different methods have been presented by several groups that oxygen reduction (ORR) is important secondary cathodic reaction during Mg corrosion [1-8]. For model galvanic couple Al-Cu-Mg, oxygen consumption on the surface of Mg along with that on Cu was observed by using oxygen sensing microoptode [1]. A simultaneous measurement of local current density by scanning vibrating electrode technique (SVET) and local oxygen concentration by oxygen sensing microoptode found a strong oxygen consumption at the active corrosion sites on the surface of commercially pure Mg in NaCl electrolyte [2]. Then, the evidence for the varying contributions of ORR to the total cathodic process on Mg alloys during atmospheric and aqueous corrosion was presented based on newly developed respirometric methods [3-5]. While around 10% of the total cathodic current was ascribed to ORR during aqueous corrosion, up to 60% of the total corrosion process was shown to be due to the ORR contribution during simulated atmospheric corrosion with wet-dry cycles. Nanoparticle-based O2 imaging was used to map decreased O2 concentration at the interface of several Mg alloys exposed to simulated body fluids [6].
High ORR rate on a slowly corroding ultra-high-purity Mg (UHP-Mg) in NaCl electrolyte was shown in our recent work [7]. Comparing that with lower ORR rate for faster corroding commercial purity Mg (CP-Mg), the rate of ORR was found to depend on the growth rate of Mg(OH)2 , which is the main corrosion product of Mg in the simple saline solution. The formation of Mg(OH)2 impeded the diffusion of oxygen from bulk electrolyte to metal interface, preventing metallic Mg substrate from the interaction with oxygen. The distribution of local concentration of dissolved gaseous H2 and O2 assessed by amperometric Clark-type micorprobes at the interface of UHP-Mg and CP-Mg demonstrated the inverse relationship between HER and ORR, and supports the finding that diffusion-controlled ORR highly depends on the barrier property of corrosion products on the surface [8]. The contribution of ORR current to total cathodic current reached 29.1% for UHP-Mg, while only 0.9% for CP-Mg after 1 hour of immersion in NaCl solution. The contribution of ORR decreases with immersion time, due to the impeded access of dissolved O2 to Mg interface caused by thickening of Mg(OH)2 layer. A numerical model was developed considering the mixed potential diagram and measured local oxygen concentration.
REFERENCES:
[1] D. Snihirova, M. Taryba, S.V. Lamaka, M.F. Montemor, Corrosion inhibition synergies on a model Al-Cu-Mg sample studied by localized scanning electrochemical techniques, Corrosion Science, 112 (2016) 408-417.
[2] E.L. Silva, S.V. Lamaka, D. Mei, M.L. Zheludkevich, The Reduction of Dissolved Oxygen During Magnesium Corrosion, ChemistryOpen, 7 (2018) 664-668.
[3] M. Strebl, S. Virtanen, Real-Time Monitoring of Atmospheric Magnesium Alloy Corrosion, Journal of The Electrochemical Society, 166 (2019) C3001-C3009.
[4] M. Strebl, M. Bruns, S. Virtanen, Editors' Choice—Respirometric in Situ Methods for Real-Time Monitoring of Corrosion Rates: Part I. Atmospheric Corrosion, Journal of The Electrochemical Society, 167 (2020) 021510.
[5] M. Strebl, M.P. Bruns, G. Schulze, S. Virtanen, Respirometric In Situ Methods for Real-Time Monitoring of Corrosion Rates: Part II. Immersion, Journal of the Electrochemical Society, (2021).
[6] B. Zeller-Plumhoff, A.R. Akkineni, H. Helmholz, D. Orlov, M. Mosshammer, M. Kühl, R. Willumeit-Römer, M. Gelinsky, Oxygen-sensitive nanoparticles reveal the spatiotemporal dynamics of oxygen reduction during magnesium implant biodegradation, NPJ Materials Degradation, 6 (2022) 95.
[7] C. Wang, D. Mei, G. Wiese, L.Q. Wang, M. Deng, S.V. Lamaka, M.L. Zheludkevich, High rate oxygen reduction reaction during corrosion of ultra-high-purity magnesium, NPJ Materials Degradation, 4 (2020) 42.
[8] C. Wang, W. Xu, D. Höche, M.L. Zheludkevich, S.V. Lamaka, Exploring the contribution of oxygen reduction reaction to Mg corrosion by modeling assisted local analysis, Journal of Magnesium and Alloys, 11 (2023) 100-109.
ACKNOWLEDGEMENTS: C. Wang, Wen Xu, thank China Scholarship Council for the funding, grants no. 201806310128 and 201908510177
Bing-Ang Mei and Laurent Pilon 2017 Meet. Abstr. MA2017-01 1468
Electric double layer capacitors (EDLCs) store energy via ion adsorption in the electric double layer forming at the electrode/electrolyte interfaces. This charge storage mechanism is very fast and highly reversible resulting in large power density and long cycle life. Applications range from regenerative breaking for hybrid electric vehicles to the smart grid. Electrochemical impedance spectroscopy (EIS) has been used widely to determine the properties of the electrode and electrolyte materials for EDLCs. The impedance of an EDLC half-cell can be measured by applying a low-amplitude sinusoidal voltage to a steady-state potential and divide the imposed potential by the output sinusoidal current. Nyquist plots showing the imaginary part as a function of the real part of the impedance is informative to determine the properties including electrode and electrolyte resistance, charge transfer or polarization resistance at the electrode/electrolyte interface, and capacitance of the half-cell. However, the interpretation of Nyquist plots has been a "subject of controversy", with no clear relations between the shape in high, intermediate, and low frequency regime of the plot and a particular property of the device.
This study aims to derive guidelines for characterization of EDLC electrode and electrolyte materials from EIS measurements. To do so, Nyquist plots from EIC measurements were numerically reproduced for EDLC half-cells with planar electrodes in aqueous TEABF4 electrolyte. The continuum model used was based on the modified Poisson-Nernst-Planck model and accounted for (i) the Stern layer at the electrode/electrolyte interface, (ii) finite ion size, and (iii) binary and symmetric electrolyte. The effect of the electrode and electrolyte resistance, charge transfer or polarization resistance at the electrode/electrolyte interface, and the capacitances were separated numerically. This was done by varying the bulk ion concentration, electrolyte length, ion diffusion coefficient in the electrolyte, and/or the magnitude of steady-state potential. Therefore, the behaviour in each frequency regime in Nyquist plots that corresponded to a particular property was clarified.
Fredrick Omenya et al 2018 Meet. Abstr. MA2018-01 531
LiCoO2 has conquered the cathode market for lithium ion batteries; recently the mixed transition metal oxide lithium nickel manganese cobalt dioxide have gained attention due to their superior electrochemical properties, better thermal stability, and lower cost. Nickel-rich compositions of lithium nickel manganese cobalt oxide (NMC) have been developed as promising high-energy, high-voltage and improved rate cathode materials capable of significantly increasing the energy density of lithium-ion batteries.
Here we report a systematic comparative study on rate capability, charge discharge behavior and nickel/lithium mixing of the nickel-rich layered oxides, NMC622, NMC811 and NCA. The results showed that both NMC622 and NMC811 have pure hexagonal layered phase with Ni/Li mixing below 4%. Both samples have similar specific capacity of about 220 mAh/g at current densities, C/10; however, NMC811 demonstrated superior electrochemical performance compared to NMC611 at higher rates. The 622 sample showed a strong dependence on the electrode loading, higher electrode loading above 15 mg/cm2 had poor performance compared to lower loading at the same current density in mA/cm2 while NMC811 sample does not show such loading dependence. This work was supported by the DOE-EERE-Battery500 consortium.
Korbinian Huber et al 2023 Meet. Abstr. MA2023-01 539
Lithium-Ion Battery electrode manufacturing is a cost- and energy-intensive process that usually relies on the use of a hazardous and expensive solvent, N-methyl-2-pyrrolidone (NMP), for cathodes. After coating the battery-slurry to a current collector, the solvent needs to be evaporated to obtain a porous electrode suitable for the use in a Lithium-Ion Battery. The solvent is a processing material and unwanted in the final product. In fact, solvent residues can fuel parasitic side-reactions within the battery cell and hence deteriorate battery lifetime and safety. The utilization of NMP in battery production plants demands costly labor protection and explosion safety measurements during mixing and coating and necessitates a highly energy and floor-space demanding drying step using thermic drying ovens of up to 100 meters.[1] For ecological and economic reasons, NMP is not emitted in large-scale production facilities, but condensed and recycled by distillation. The NMP-recovery system adds additional floor space and energy demand to the production site. The drying and solvent recovery can account for up to 39% of the total energy demand of a Lithium-Ion Battery Cell production and therefore produce significant CO2 emissions.[2] A solvent-free, dry electrode manufacturing that eliminates the use of solvents hence reduces the floor space, energy demand and cost of a production plant and eases safety and environmental concerns. Different approaches have been reported in the literature to put dry coating into practice,[3],[4] yet most strategies, for example spray coating or brush coating, lack of a feasible implementation into large scale production. In contrast, a dry coating approach that is sometimes referred to as the Maxwell-Process is, according to media reports, currently installed at Tesla's Gigafactories in Berlin and Austin.[5] In fact, a publicly available teardown of a Tesla 4680 battery cell hints that a dry coated anode could already be used in commercial vehicles.[6] Compared to the state-of-the-art electrode manufacturing process, dry coating requires different polymeric binder systems that form spiderweb-like structures of fine fibrils connecting the electrochemical active particles and the conductive additive particles. PTFE is highly suitable since it is stable towards typical electrolytes and cathode materials and is known to easily form fibrils. These properties are also utilized to produce expanded PTFE membranes like Gore-Tex. The fibril-network in the battery context is primarily formed by a high-shear dry mixing process. The powder mixture is then compressed to a free-standing electrode film (powder-to-film) by a heated rolling mill (calender) and the resulting film is ultimately laminated to a current collector foil (film-to-foil). We demonstrate that a twin screw-extruder can be used to tune the degree of binder-fibrillation during dry-mixing of NMC-based cathode mixtures. Extrusion based mixing allows to use powder mixtures with little amounts of PTFE binder (1 wt%) to produce self-supporting, free-standing cathode films and, ultimately, battery electrodes with high flexibility and sufficient mechanical stability. We show a superior rate-performance and cycling-stability of single-layer pouch cells with dry-coated cathodes compared to cells with wet coated reference electrodes of likewise composition and electrode design. Possible causes for the improved performance focusing on microstructural electrode properties are discussed.
[1] Westphal, Bastian G.; Kwade, Arno (2018): Critical electrode properties and drying conditions causing component segregation in graphitic anodes for lithium-ion batteries. In: Journal of Energy Storage 18, S. 509–517.
[2] Erik Emilsson, Lisbeth Dahllöf (2019): Lithium-Ion Vehicle Battery Production. IVL Swedish Environmental Research Institute.
[3] Duffner, Fabian; Kronemeyer, Niklas; Tübke, Jens; Leker, Jens; Winter, Martin; Schmuch, Richard (2021): Post-lithium-ion battery cell production and its compatibility with lithium-ion cell production infrastructure. In: Nat Energy 6 (2), S. 123–134.
[4] Verdier, Nina; Foran, Gabrielle; Lepage, David; Prébé, Arnaud; Aymé-Perrot, David; Dollé, Mickaël (2021): Challenges in Solvent-Free Methods for Manufacturing Electrodes and Electrolytes for Lithium-Based Batteries. In: Polymers 13 (3).
[5] https://www.tagesspiegel.de/berlin/tesla-coup-mit-giga-berlin-neue-technologie-soll-wasserverbrauch-minimieren/27244032.html
[6] https://www.youtube.com/watch?v=8WPPBhqeekw

Figure 1
E. Joseph Nemanick et al 2016 Meet. Abstr. MA2016-02 897
Cell reversal in lithium ion (Li-ion) batteries is the condition of the anode electrochemical potential rising above that of the cathode, resulting in a negative voltage measured at the cell level. There are two primary reactions that occur at the anode at high potentials which increase cell impedance: oxidation of copper current collector, and oxidization of the carbonate electrolytes to CO2. At the cathode, the reducing potential can lead to the electrodeposition of copper to form dendrites, which pose a shorting risk if they bridge the anode and cathode. Cell reversal can be caused by poorly matched cells, a failure of the battery management electronics, or a defective cell in a pack. Under these conditions, one or several of the cells can go into reversal causing performance decreases or even a dangerous thermal runaway event. This paper examines a pack of commercial 18650 Li-ion cells in simulated geosynchronous orbit (GEO) test under conditions where one or more cells were forced into reversal.
Panasonic B cells were matched and assembled into a pack to create a virtual cell. Measurements were collected using matched current shunts, and cells were cycled at a 60% DOD, but with only 90% of removed charge replaced each cycle to simulate a battery management system failure. After failure, cells were non-destructively examined using CT X-ray, and then dissected for failure analysis.
The parallel cell pack operated far into reversal, with up to four complete cycles (Fig. 1) completed before all cells shorted. CT X-ray scans of the cells after shorting detectable amounts of copper dendrites in the cell, and demonstrated the first nondestructive test for cell reversal in Li-ion (Fig. 2). Destructive physical analysis of the cells showed extensive copper corrosion at the anode current collector as well as copper dendrites that were found to have fully penetrated the separator in selected areas.
These results show that these cells can operate several cycles into deep reversal without going into thermal runaway, despite the observed growth of copper dendrites which pierce the cell separator. This has strong implications on cell safety and battery management.
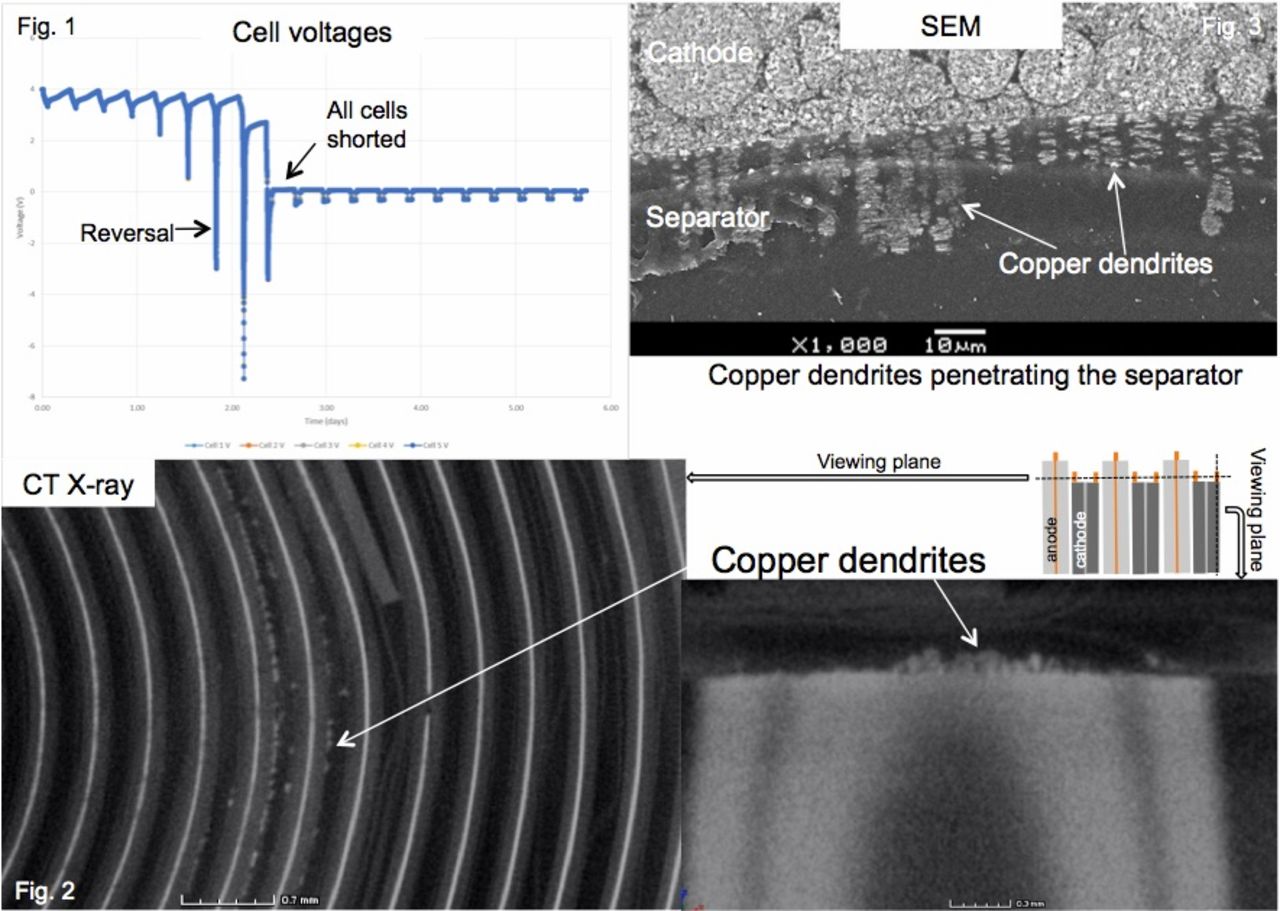
Figure 1
Honghe Zheng et al 2010 Meet. Abstr. MA2010-02 200
Abstract not Available.
Won young Choi et al 2023 Meet. Abstr. MA2023-02 1934
Polymer electrolyte fuel cells (PEFCs) have emerged as one of the most promising next-generation energy devices for both passenger-owned and heavy-duty vehicles due to their high energy efficiency, low emissions, and quiet operation. However, the performance of the fuel cell electrode is often limited by the strong specific adsorption of long sidechain (LSC) perfluoro-sulfonic acid (PFSA) ionomer onto the platinum (Pt) catalyst2, which results in a decrease in the electrocatalytic activity1 as well as increase oxygen mass transport resistenace at the MEA level. To address this issue, a short sidechain (SSC) ionomer has been developed, which has been found to exhibit excellent efficiency over LSC ionomer3 The reduced specific adsorption of SSC ionomer onto the Pt catalyst results in a higher available surface area for the electrochemical reaction, which improves the overall fuel cell performance. The sidechain length of ionomers plays a crucial role in determining the overall cell performance4 however, due to the differences between them, the interactions among slurry components are not yet fully understood.
Unlike the previous literatures, we investigates the details of ionomer sidechain length and its impact on the interaction among slurry components and resulting electrode microstructure, through a comparative analysis of LSC and SSC ionomers.5 Our hypothesis propose that the physical interactions between components are considerably impacted by the sidechains with a specific emphasis on the mobility and ion-pair association in the slurry. To probe the influence of ionomer sidechains, we conducted a thorough investigation wherein incrementally varied its concentration from 0 to 1.5 mmolSO3-gC-1, at intervals of 0.3 mmolSO3-gC-1. The main purpose of this evaluation was to observe the evolution of particle aggregation according to the correlation between adsorbed ionomer and free ionomer in electrode slurry. Drawing from the U-shaped viscosity profile obtained through rheological measurements, we propose an optimized ionomer concentration of 0.6 mmolSO3-gC-1 for the SSC ionomer-based slurry and 0.9 mmolSO3-gC-1 for the LSC ionomer-based slurry. To characterize the microstructure of the as-prepared electrode, we employed scanning electron microscopy (SEM) and confocal microscopy, with a focus on particle agglomeration and the homogeneous ionomer distribution. Finally, the electrochemical analysis was performed on the fabricated membrane electrode assembly (MEA) in a 25 cm² single cell for a cathode Pt loading of 0.1 mgPtcm⁻². As a result, the optimized SSC ionomer-based electrode exhibited low local O₂ transport resistance and high Pt utilization, leading to over a 40 % enhancement in fuel cell performance at 0.6 V with a high proton accessibility value above 0.85 when compared to the optimized LSC ionomer-based electrode. This study highlights the crucial importance of understanding the effects of ionomer sidechain length on electrode slurry interactions and microstructure and establishes a significant correlation between them. Our findings contribute to the ongoing development of high-performance PEFCs, offering valuable insights into optimizing electrode composition and structure for enhanced fuel cell performance.
(1) Ahn, C.-Y.; Park, J. E.; Kim, S.; Kim, O.-H.; Hwang, W.; Her, M.; Kang, S. Y.; Park, S.; Kwon, O. J.; Park, H. S. Differences in the electrochemical performance of Pt-based catalysts used for polymer electrolyte membrane fuel cells in liquid half-and full-cells. Chemical Reviews 2021, 121 (24), 15075-15140.
(2)Kodama, K.; Motobayashi, K.; Shinohara, A.; Hasegawa, N.; Kudo, K.; Jinnouchi, R.; Osawa, M.; Morimoto, Y. Effect of the side-chain structure of perfluoro-sulfonic acid ionomers on the oxygen reduction reaction on the surface of Pt. ACS Catalysis 2018, 8 (1), 694-700. (2) Garsany, Y.; Atkinson, R. W.; Sassin, M. B.; Hjelm, R. M.; (3) Gould, B. D.; Swider-Lyons, K. E. Improving PEMFC Performance Using Short-Side-Chain Low-Equivalent-Weight PFSA Ionomer in the Cathode Catalyst Layer. Journal of The Electrochemical Society 2018, 165 (5), F381-F391.
(4) Ramaswamy, N.; Kumaraguru, S.; Koestner, R.; Fuller, T.; Gu, W.; Kariuki, N.; Myers, D.; Dudenas, P. J.; Kusoglu, A. Editors' Choice—Ionomer Side Chain Length and Equivalent Weight Impact on High Current Density Transport Resistances in PEMFC Cathodes. Journal of The Electrochemical Society 2021, 168 (2), 024518.
(5) Mauritz, K. A.; Moore, R. B. State of Understanding of Nafion. Chemical Reviews 2004, 104 (10), 4535-4586. DOI: 10.1021/cr0207123.
Naga Siva Gunda et al 2018 Meet. Abstr. MA2018-02 1997
Emerging trends in Artificial Intelligence (AI) have provided a path to make better predictions in a variety of fields, including the detection of medical diseases, weather patterns, water, food quality patterns, remote sensing etc. Deep learning, a branch of AI that uses deep convolutional neural networks (CNN) modeled after the human brain, have been prominently used for accurate identification of facial features, text, and voice. This technique is used predominantly to classify a set of objects, that may not necessarily have a fixed set of features, making it very difficult to detect programmatically. As an example, researchers have started implementing deep learning techniques to identify non-uniform cancer cells from the high-resolution microscopic images of tissue samples. Similarly, most of the current screening/diagnostic devices are color-based indicators. By looking at the amount of color appearance on sensor zones of such devices, one can identify the level of certain contaminants, diseases or infections. Typically, look-up tables will be provided to classify the level of sensing parameters based on color intensity. This is a task that would be extremely time consuming and challenging to do physically, given that a complete database mapping color to concentration would have to be created and a minor difference may or may not indicate a significant concentration change. Deep learning will be very helpful for accurate identification of color and its intensity on such diagnostic devices. In the present work, we have developed an AI-based mobile application platform, that can capture the sensor image using an inbuilt smartphone camera, identify the presence of sensing parameter and classify the level of sensing parameter based on color intensity identified in the training sets on the captured image using deep CNN algorithm.
As a test case, we have implemented the developed AI-based mobile application platform for water quality monitoring for bacterial contamination. We used a low-cost rapid test kit i.e., Mobile Water Kit (MWK), developed by Gunda et al. [Anal. Methods, 2014, 6, 6236-6246 and Analyst, 2016, 141, 2920-2929] for monitoring the quality of water for bacterial contamination. MWK detects indicator bacteria (E. coli) in water samples within an hour, based on the appearance of pinkish red color on the surface of the sensing area. The color intensity represents the level of bacteria in water samples. Using the AI-based mobile app, we capture the image of the MWK sensing area (after testing water samples) and classify them into E. coli present images (i.e. E. coli images) and E. coli absent images (non-E. coli images). Deep learning works very well when there is an abundance of training data and there are certain factors that will make it difficult to programmatically distinguish between types. Using traditional computer vision techniques, one would scan the colors of each concentration. However, determining the color intensity for each concentration level is very difficult (especially because these are different shades of pinkish red for MWK). Using deep learning, this is made easy as the system determines these color intensities through training sets that have been provided statistically.
In this present work, we have collected training data from MWK by testing the water samples with known concentrations of E. coli bacteria and then manually segregated the captured images based on whether the sample contains E. coli or not. Then we wrote a labeling script to label these images based on their type. We then used Google Tensorflow (a deep learning Artificial Intelligence open source tool) to distinguish between E. coli and non-E. coli images. Subsequently, we used the labeling script to classify whether an MWK tested image contains E. coli or not. As of now, we can classify the images with approximately 99% accuracy. We will also able to predict concentration levels using this method.
Jin Tang et al 2017 Meet. Abstr. MA2017-01 441
In recent years, the lithium iron phosphate battery is widely used in the fields of electric vehicles and energy storage because of its high energy density, long cycle life and safety[1], but the existing battery technology was not enough to meet the requirements of electric vehicles[2]. So it is of great importance to research performances of battery.
In this paper, the influence of different depth of discharge (DOD) on the cycle life of the battery was investigated. The specific research process is as follows, three kinds of LiFePO4 batteries of the same type were charged and discharged at three different discharge depths (30% DOD, 50% DOD and 100% DOD) under constant conditions of 40℃and 1C (1.3A), and the discharge capacity decay curve and decay rate curve were measured after a certain number of cycles.
Discharge capacity decay curve and decay rate curve measured under different discharge depth are shown in Figure 1. As can be seen from the left graphic, the discharge capacity of the battery will have a slight increase in the early, this is because the battery anode material has not been fully activated during the initial cycle, as the cycle progresses, the electrolyte gradually penetrates into the interior of the electrode material, and the lithium ions smoothly migrate to the inside of the electrode material and undergo reversible deintercalation reaction, resulting in an increase in the capacity of the battery. In addition, At the beginning of the cycle, the depth of discharge has little effect on the capacity of the three groups of batteries. When the cycle continues, the discharge capacity of the LiFePO4 battery gradually decreased, the attenuation of battery capacity by the depth of discharge is more and more obvious. The right capacity fading rate curve shows that battery capacity decay rate remained the same at the beginning of the cycle. At this time, the influence of the battery capacity by depth of discharge is almost independent. After the initial cycle, the deeper the depth of discharge, the faster the cell capacity decays, and there is a significant the positive correlation between the depth of discharge and the decay rate of battery capacity. Due to the different depth of discharge, the internal structure of the electrode material will occur to different degrees of deterioration.
Fig. 1 the discharge capacity decay curve and decay rate curve under different discharge depth.
It can be seen from the above studies that the effect of the battery cycle life by depth of discharge is various in different cycle stages. In the early cycle, LiFePO4 battery capacity at different depth of discharge changes in the same law, indicating that the depth of discharge has no effect on the battery life in the early cycle. But as the cycle continues, the greater depth of discharge, the faster decay of battery capacity, the battery cycle life decline faster.
Acknowledgements
This work is financially supported by the National High Technology Research and Development Program of China (863Program, no.2015BAG01B01).
References
[1] Forgez C, Vinh Do D, Friedrich G, et al. Thermal modeling of a cylindrical LiFePO4/graphite lithium-ion battery[J]. Journal of Power Sources, 2010,195(9):2961-2968.
[2] Ritchie A, Howard W. Recent developments and likely advances in lithium-ion batteries[J]. Journal of Power Sources, 2006,162(2):809-812.

Figure 1
Open all abstracts, in this tab
Nowduru Ravikiran et al 2023 Meet. Abstr. MA2023-02 3233
Mono-layer graphene has shown path-breaking potential in almost all areas that are useful to humankind. However, research in the last decade indicated that few-layer graphene (FLG) (3-10 layers) and multi-layer graphene (MLG) (>10 layers) could also be utilized for a wide range of applications. The synthesis of mono-layer graphene at a large scale is quite complex. Producing FLG and MLG at a kilogram scale is equally challenging. A vast amount of literature on different routes to prepare FLG and MLG is available. However, most of the preparation routes involve the use of hazardous chemicals, copious amounts of water, and thermal treatments and are time taking. Also, these processes need bigger machinery to convert the lab-scale methods into continuous large-scale production. On the contrary, in this work we report a novel process of producing FLG and MLG along with trace quantities of graphene (< 3 layers) developed using a kitchen mixer and a modified mixing jar (3 litres). The highlights of this process include the use of no hazardous chemicals/solvents, no washing or thermal treatment needed, much less time (30 minutes), and large quantity (250-500 grams) per batch. Our study successfully converted natural graphite flakes (NGF) into FLG and MLG by carrying out rapid liquid phase shear mixing using a green solvent. The shear mixing and the green solvent convert the NGF into FLG and MLG. The quantity of FLG and MLG getting produced is time-dependent. It has been optimized by thoroughly characterizing FLG and MLG using techniques like particle size analyser, X-ray diffraction, Raman spectroscopy, and electron microscopy. The conversion of NGF to FLG and MLG is also affirmed by studying changes in properties using tools like thermogravimetry, BET, and UV-visible spectroscopy. The developed process is rapid, efficient, economical, scalable, continuous, and, most importantly, chemical free.
The graphenaceous powder produced unlike graphite can be easily coated on different substrates using appropriate binders and thus makes it useable for energy storage applications. Also the fine particle size of graphite makes is an ideal material to be used as lubricant, both in solid and liquid mediums. The developed material can be explored for its applications in several other areas which require FLG/MLG.
Ya-Ping Sun 2023 Meet. Abstr. MA2023-02 3273
In the vast research field on carbon nanostructures, a seemingly broadly accepted family configuration of nanoscale carbon allotropes is such that fullerenes take the zero-dimensional dot spot, in perfect harmony with one-dimensional carbon nanotubes and two-dimensional graphene nanosheets. More recently, however, a threat to the nice "family value" has been the emergence of small carbon nanoparticles, which are carbon particles of a few nanometers in size without any defined crystal structures or largely amorphous and populated with abundant surface and other defects. The growing experimental evidence has revealed the unique and/or advantageous optical and photoexcited state properties and photoinduced redox characteristics of the small carbon nanoparticles, especially the spectacular performance enhancements upon their being effectively surface passivated by organic species for the hard-core/soft-shell nanostructures defined as carbon dots. Thus, the ugly and dirty dot-like carbon particles have been elevated to the position of being able to compete with pretty and perfect fullerenes, which by the way are nano-sized molecules strictly speaking, for the title of nanoscale carbon allotrope at the zero-dimension. Nevertheless, carbon dots are not short of controversies, which are due largely to the fact that samples sold or labeled as "carbon dots" in store or literature reports, respectively, are often not what they are advertized, but complex mixtures of varying structures and compositions. Some of these will be discussed in the presentation, so are challenges and opportunities in this seemingly rapid expanding research field.

Figure 1
Penghui Zhu et al 2023 Meet. Abstr. MA2023-02 100
The development of high areal energy density electrodes with layered metal oxide cathode materials is currently a hot topic in research and industry. However, for automotive applications, current research focuses on the merger of two concepts: (i) the "thick-film concept" which enables a high energy density due to a reduced amount of inactive materials, and (ii) the "three-dimensional (3D) battery concept", which provides a high power density with improved interfacial kinetics at mass areal capacity ≥ 6.5 mAh/cm2. Latter could be realized by applying ultrafast laser patterning of electrodes which in turn includes an advanced 3D electrode design. Briefly, a rapid and homogeneous electrode wetting with liquid electrolyte can be induced, and besides a high capacity retention during long-term cycling. However, the mass loss due to laser patterning needs to be taken into account, since the cathode represents about 50 % of the total material costs of LIBs. Thus, the use of electrode structures with a high aspect ratio as well as a significantly reduced material removal is of great importance. Besides, cost reduction and environmentally friendly production by applying aqueous processing of cathode has been intensively investigated recently. In this work, 150 µm thick-film Li(Ni0.6Mn0.2Co0.2)O2 electrodes were manufactured with both water-based binders and PVDF binder by tape-casting. Both cathodes are anodes were subsequently laser patterned to achieve 3D architectures. Finally, the NMC 622 cathodes were assembled in pouch cells versus graphite anodes for studying the impact of laser patterning and binders on the electrochemical performance by applying rate capability analysis, electrochemical impedance spectroscopy (EIS), and lifetime analysis.
Jorge Antonio Cardenas et al 2023 Meet. Abstr. MA2023-02 101
Additive manufacturing techniques can enable the fabrication of batteries in nonconventional form factors, enabling higher practical energy densities due to improved power source packing efficiency. Furthermore, energy density can be improved by transitioning from conventional Li-ion materials to lithium metal anodes and conversion cathodes. Iron disulfide (FeS2) and iron trifluoride (FeF3) are two promising conversion cathodes of commercial and academic interest, but the 3D printing of inks made from these materials for custom-form battery applications has yet to be demonstrated. In this work, the deposition of FeS2 and FeF3 inks are investigated and optimized using direct-ink-write (DIW) 3D printing, in addition to the development of printable separators and packages to produce custom-form batteries. Two distinct custom form-factors, one on wave-shaped current collectors and the other on cylindrical rod current collectors, are demonstrated and shown to exhibit performance similar to coin cells when conventional Celgard separators are used. Additionally, FeF3 cells were integrated with a DIW printed separator consisting of an electrolyte exchanged PVDF-HFP based ionogel [1]. In the case of FeS2, it was found that cathodes with a ridged surface, produced from the filamentary extrusion of highly concentrated inks (60-70% solids w/w%) exhibited optimal power, uniformity, and stability [2]. Finally, progress toward fully-printing custom-form batteries using metal powder bed printed cases and stereolithographically printed gaskets is demonstrated. Overall, the additive manufacturing of conversion electrodes, separators, and battery packaging is shown to be a viable path toward the making of custom-form cells. More broadly, electrode ridging is found to optimize rate capability, a finding that may have broad impact beyond FeS2, FeF3 and additive manufacturing.
[1] A.S. Lapp, L.C. Merrill, B.R. Wygant, D.S. Ashby, A.S. Bhandarkar, A. Zhang, E.J. Fuller, K.L. Harrison, T.N. Lambert, and A.A. Talin. Room Temperature Pseudo-Solid State Iron Fluoride Conversion Battery with High Ionic Conductivity. ACS Applied Materials & Interfaces 15, 893-902, 2022.
[2] J.A. Cardenas, J.P. Bullivant, I.V. Kolesnichenko, D.J. Roach, M.A. Gallegos, E.N. Coker, T.N. Lambert, E. Allcorn, A.A. Talin, A.W. Cook, and K.L. Harrison. 3D Printing of Ridged FeS2 Cathodes for Improved Rate Capability and Custom-Form Lithium Batteries. ACS Applied Materials & Interfaces 14, 45342-45351. 2022.
Alexander Thomas et al 2023 Meet. Abstr. MA2023-02 104
Facing the challenges of degradation processes in sodium metal batteries, a lot of new anode materials were prepared and tested. Especially Hard Carbon is a promising candidate for sodium ion batteries.[1] However, the competitive battery performance as gravimetric and volumetric energy density cannot be achieved without metallic sodium as an anode.[2]
Another way to stabilize a sodium metal battery is the formation of an artificial solid-electrolyte interface (SEI). In the recent years, a lot of efforts were devoted for development of organic and inorganic protective layers. Most of the processes to form an artificial SEI are not easy for application, or result in a weak long-term stability of a sodium metal battery.[3]
Here, we can introduce a new artificial SEI, which can easily be formed on top of metallic sodium and is also stable upon a long-term cycling. The capacity retention in a full cell setup (Swagelok cell, layered sodium cobalt titanium oxide as a cathode material, 1 M NaClO4 in EC/PC as an electrolyte) is of nearly 85 % after 500 cycles with a 1C current density. A reference full cell with non-protected sodium only reaches 20 % after the same cycling conditions.
A possible explanation for the observed improvement in the cell performance was done based on comprehensive electrochemical, chemical and structural studies.
Acknowledgment
This work was supported by the Bundesministerium für Bildung und Forschung is carried out as part of the HeNa project (03XP0390C).
Additional information
Patent pending.
References
[1] D. A. Stevens, J. R. Dahn, J. Electrochem. Soc., 2000, 147(4), 127
[2] H. Wang, E. Matios, J. Luo and W. Li, Chem. Soc. Rev., 2020, 49, 3783 —3805
[3] N. Tapia-Ruiz et al., J. Phys. Energy, 2021, 3, 031503

Figure 1
Open all abstracts, in this tab
Sviatlana Lamaka et al 2023 Meet. Abstr. MA2023-02 1136
Unravelling the mechanisms of magnesium corrosion is vital for establishing reliable research methodologies, developing new alloys and predicting magnesium degradation behaviour. Hydrogen evolution reaction is widely regarded as by far the main cathodic process during corrosion of magnesium. Hence, tracing the amount of released hydrogen was thought to be a reliable measure of magnesium degradation rate. Recently, experimental evidences obtained by different methods have been presented by several groups that oxygen reduction (ORR) is important secondary cathodic reaction during Mg corrosion [1-8]. For model galvanic couple Al-Cu-Mg, oxygen consumption on the surface of Mg along with that on Cu was observed by using oxygen sensing microoptode [1]. A simultaneous measurement of local current density by scanning vibrating electrode technique (SVET) and local oxygen concentration by oxygen sensing microoptode found a strong oxygen consumption at the active corrosion sites on the surface of commercially pure Mg in NaCl electrolyte [2]. Then, the evidence for the varying contributions of ORR to the total cathodic process on Mg alloys during atmospheric and aqueous corrosion was presented based on newly developed respirometric methods [3-5]. While around 10% of the total cathodic current was ascribed to ORR during aqueous corrosion, up to 60% of the total corrosion process was shown to be due to the ORR contribution during simulated atmospheric corrosion with wet-dry cycles. Nanoparticle-based O2 imaging was used to map decreased O2 concentration at the interface of several Mg alloys exposed to simulated body fluids [6].
High ORR rate on a slowly corroding ultra-high-purity Mg (UHP-Mg) in NaCl electrolyte was shown in our recent work [7]. Comparing that with lower ORR rate for faster corroding commercial purity Mg (CP-Mg), the rate of ORR was found to depend on the growth rate of Mg(OH)2 , which is the main corrosion product of Mg in the simple saline solution. The formation of Mg(OH)2 impeded the diffusion of oxygen from bulk electrolyte to metal interface, preventing metallic Mg substrate from the interaction with oxygen. The distribution of local concentration of dissolved gaseous H2 and O2 assessed by amperometric Clark-type micorprobes at the interface of UHP-Mg and CP-Mg demonstrated the inverse relationship between HER and ORR, and supports the finding that diffusion-controlled ORR highly depends on the barrier property of corrosion products on the surface [8]. The contribution of ORR current to total cathodic current reached 29.1% for UHP-Mg, while only 0.9% for CP-Mg after 1 hour of immersion in NaCl solution. The contribution of ORR decreases with immersion time, due to the impeded access of dissolved O2 to Mg interface caused by thickening of Mg(OH)2 layer. A numerical model was developed considering the mixed potential diagram and measured local oxygen concentration.
REFERENCES:
[1] D. Snihirova, M. Taryba, S.V. Lamaka, M.F. Montemor, Corrosion inhibition synergies on a model Al-Cu-Mg sample studied by localized scanning electrochemical techniques, Corrosion Science, 112 (2016) 408-417.
[2] E.L. Silva, S.V. Lamaka, D. Mei, M.L. Zheludkevich, The Reduction of Dissolved Oxygen During Magnesium Corrosion, ChemistryOpen, 7 (2018) 664-668.
[3] M. Strebl, S. Virtanen, Real-Time Monitoring of Atmospheric Magnesium Alloy Corrosion, Journal of The Electrochemical Society, 166 (2019) C3001-C3009.
[4] M. Strebl, M. Bruns, S. Virtanen, Editors' Choice—Respirometric in Situ Methods for Real-Time Monitoring of Corrosion Rates: Part I. Atmospheric Corrosion, Journal of The Electrochemical Society, 167 (2020) 021510.
[5] M. Strebl, M.P. Bruns, G. Schulze, S. Virtanen, Respirometric In Situ Methods for Real-Time Monitoring of Corrosion Rates: Part II. Immersion, Journal of the Electrochemical Society, (2021).
[6] B. Zeller-Plumhoff, A.R. Akkineni, H. Helmholz, D. Orlov, M. Mosshammer, M. Kühl, R. Willumeit-Römer, M. Gelinsky, Oxygen-sensitive nanoparticles reveal the spatiotemporal dynamics of oxygen reduction during magnesium implant biodegradation, NPJ Materials Degradation, 6 (2022) 95.
[7] C. Wang, D. Mei, G. Wiese, L.Q. Wang, M. Deng, S.V. Lamaka, M.L. Zheludkevich, High rate oxygen reduction reaction during corrosion of ultra-high-purity magnesium, NPJ Materials Degradation, 4 (2020) 42.
[8] C. Wang, W. Xu, D. Höche, M.L. Zheludkevich, S.V. Lamaka, Exploring the contribution of oxygen reduction reaction to Mg corrosion by modeling assisted local analysis, Journal of Magnesium and Alloys, 11 (2023) 100-109.
ACKNOWLEDGEMENTS: C. Wang, Wen Xu, thank China Scholarship Council for the funding, grants no. 201806310128 and 201908510177
Won young Choi et al 2023 Meet. Abstr. MA2023-02 1934
Polymer electrolyte fuel cells (PEFCs) have emerged as one of the most promising next-generation energy devices for both passenger-owned and heavy-duty vehicles due to their high energy efficiency, low emissions, and quiet operation. However, the performance of the fuel cell electrode is often limited by the strong specific adsorption of long sidechain (LSC) perfluoro-sulfonic acid (PFSA) ionomer onto the platinum (Pt) catalyst2, which results in a decrease in the electrocatalytic activity1 as well as increase oxygen mass transport resistenace at the MEA level. To address this issue, a short sidechain (SSC) ionomer has been developed, which has been found to exhibit excellent efficiency over LSC ionomer3 The reduced specific adsorption of SSC ionomer onto the Pt catalyst results in a higher available surface area for the electrochemical reaction, which improves the overall fuel cell performance. The sidechain length of ionomers plays a crucial role in determining the overall cell performance4 however, due to the differences between them, the interactions among slurry components are not yet fully understood.
Unlike the previous literatures, we investigates the details of ionomer sidechain length and its impact on the interaction among slurry components and resulting electrode microstructure, through a comparative analysis of LSC and SSC ionomers.5 Our hypothesis propose that the physical interactions between components are considerably impacted by the sidechains with a specific emphasis on the mobility and ion-pair association in the slurry. To probe the influence of ionomer sidechains, we conducted a thorough investigation wherein incrementally varied its concentration from 0 to 1.5 mmolSO3-gC-1, at intervals of 0.3 mmolSO3-gC-1. The main purpose of this evaluation was to observe the evolution of particle aggregation according to the correlation between adsorbed ionomer and free ionomer in electrode slurry. Drawing from the U-shaped viscosity profile obtained through rheological measurements, we propose an optimized ionomer concentration of 0.6 mmolSO3-gC-1 for the SSC ionomer-based slurry and 0.9 mmolSO3-gC-1 for the LSC ionomer-based slurry. To characterize the microstructure of the as-prepared electrode, we employed scanning electron microscopy (SEM) and confocal microscopy, with a focus on particle agglomeration and the homogeneous ionomer distribution. Finally, the electrochemical analysis was performed on the fabricated membrane electrode assembly (MEA) in a 25 cm² single cell for a cathode Pt loading of 0.1 mgPtcm⁻². As a result, the optimized SSC ionomer-based electrode exhibited low local O₂ transport resistance and high Pt utilization, leading to over a 40 % enhancement in fuel cell performance at 0.6 V with a high proton accessibility value above 0.85 when compared to the optimized LSC ionomer-based electrode. This study highlights the crucial importance of understanding the effects of ionomer sidechain length on electrode slurry interactions and microstructure and establishes a significant correlation between them. Our findings contribute to the ongoing development of high-performance PEFCs, offering valuable insights into optimizing electrode composition and structure for enhanced fuel cell performance.
(1) Ahn, C.-Y.; Park, J. E.; Kim, S.; Kim, O.-H.; Hwang, W.; Her, M.; Kang, S. Y.; Park, S.; Kwon, O. J.; Park, H. S. Differences in the electrochemical performance of Pt-based catalysts used for polymer electrolyte membrane fuel cells in liquid half-and full-cells. Chemical Reviews 2021, 121 (24), 15075-15140.
(2)Kodama, K.; Motobayashi, K.; Shinohara, A.; Hasegawa, N.; Kudo, K.; Jinnouchi, R.; Osawa, M.; Morimoto, Y. Effect of the side-chain structure of perfluoro-sulfonic acid ionomers on the oxygen reduction reaction on the surface of Pt. ACS Catalysis 2018, 8 (1), 694-700. (2) Garsany, Y.; Atkinson, R. W.; Sassin, M. B.; Hjelm, R. M.; (3) Gould, B. D.; Swider-Lyons, K. E. Improving PEMFC Performance Using Short-Side-Chain Low-Equivalent-Weight PFSA Ionomer in the Cathode Catalyst Layer. Journal of The Electrochemical Society 2018, 165 (5), F381-F391.
(4) Ramaswamy, N.; Kumaraguru, S.; Koestner, R.; Fuller, T.; Gu, W.; Kariuki, N.; Myers, D.; Dudenas, P. J.; Kusoglu, A. Editors' Choice—Ionomer Side Chain Length and Equivalent Weight Impact on High Current Density Transport Resistances in PEMFC Cathodes. Journal of The Electrochemical Society 2021, 168 (2), 024518.
(5) Mauritz, K. A.; Moore, R. B. State of Understanding of Nafion. Chemical Reviews 2004, 104 (10), 4535-4586. DOI: 10.1021/cr0207123.
Sannakaisa Virtanen 2022 Meet. Abstr. MA2022-02 750
Recently, we have introduced novel respirometric methods for real-time monitoring of corrosion, both for atmospheric corrosion [1,2] and immersion conditions [3]. The approach is based on monitoring the rate of the cathodic reactions with help of different types of sensors; this enables to track the O2 reduction reaction and the H2 evolution reaction simultaneously. Different experimental setups have been developed, from static to flow cell scenarios. Moreover, combining respirometry with time-lapse microscopy reveals interesting details on initiation and propagation of corrosion. Most recently, the respirometric setups were coupled with electrochemistry [4], enlarging the corrosion cases to be studied from open-circuit conditions to potential-controlled systems. With this, it is possible for instance to follow and quantify the amount of oxygen evolving during transpassive dissolution of stainless steels at potentials above the water stability region (i.e., the net electrical current/charge can be assigned into partial currents for oxygen evolution and for metal dissolution). Examples will be presented, to illustrate the potential of these techniques to reveal new insights into corrosion mechanisms of active and passive metals and alloys.
References
M. Strebl, S. Virtanen, Real-Time Monitoring of Atmospheric Magnesium Alloy Corrosion, Journal of The Electrochemical Society 166 (2019) C3001-C3009
M. Strebl, M. Bruns, S. Virtanen, Editors' Choice—Respirometric in Situ Methods for Real-Time Monitoring of Corrosion Rates: Part I. Atmospheric Corrosion, J. Electrochem. Soc. 167 (2020) 21510
M.G. Strebl, M.P. Bruns, G. Schulze, S. Virtanen, Respirometric In Situ Methods for Real-Time Monitoring of Corrosion Rates: Part II. Immersion, J. Electrochem. Soc. 168 (2021) 11502
M.G. Strebl, M.P. Bruns, S. Virtanen, Coupling Respirometric HER and ORR Monitoring with Electrochemical Measurements, Electrochimica Acta 412 (2022) 140152
Shohei Ogawa et al 2019 Meet. Abstr. MA2019-02 1370
Optimizing the structure of the catalyst layer is critical to improving the proton exchange membrane fuel cell (PEMFC) performance at low Platinum (Pt) loading. A cell with reduced cathode Pt-loading for the cost reduction suffers from a limited performance at high current density, and it is a barrier to practical applications of PEMFC [1]. In addition to Pt-loadings, the types of carbon supports affect cell performance [2]. In the prior work, the nano-scale scanning transmission electron microscopy computed tomography (STEM-CT) was applied to visualize the individual Pt particle distributions in 3D at two Pt-loadings on two types of carbon supports [3]. This imaging showed that with medium surface area, low porosity carbon support (e.g., Vulcan), a large majority of Pt particles are on the surface of the carbon support. In contrast, STEM-CT showed that with a high-surface-area carbon (HSC), a majority of Pt particles are within the porous carbon support. These internal Pt particles are not in contact with the acidic electrolyte, therefore it is unclear how protons are transported to the Pt surface and how the oxygen reduction reaction (ORR) occurs on these Pt particles. In addition, prior experimental results suggest that the utilization of inner Pt particles is improved at high relative humidity with HSC since the micropores in the carbon support are likely to be filled with condensed water [3].
Here, to investigate the characteristics of those catalyst materials, we developed a transport and ORR model on 3D structures obtained from the STEM-CT images. We solve the Poisson-Nernst-Planck equations to model the electromigration and surface charge effects on the proton concentration in the porous carbon supports with a continuum approximation. Figure 1 shows the ORR current density on the Pt particles in the HSC support at the electrode potential, 0.7 V. The simulation result suggests that negative surface charge on Pt particles may be necessary for a high proton concentration and activity of inner particles in water-filled micropores at high relative humidity. This model can be used to characterize the performance of the catalyst layers in different configurations.
Acknowledgement
This work was partially supported by the U.S. Department of Energy, Office of Energy Efficiency and Renewable Energy under grant DE-EE0007271.
References
[1] A. Kongkanand and M. F. Mathias, "The Priority and Challenge of High-Power Performance of Low-Platinum Proton-Exchange Membrane Fuel Cells," J. Phys. Chem. Lett., vol. 7, no. 7, pp. 1127–1137, Apr. 2016.
[2] V. Yarlagadda et al., "Boosting Fuel Cell Performance with Accessible Carbon Mesopores," ACS Energy Lett., vol. 3, no. 3, pp. 618–621, 2018.
[3] E. Padgett et al., "Editors' Choice—Connecting Fuel Cell Catalyst Nanostructure and Accessibility Using Quantitative Cryo-STEM Tomography," J. Electrochem. Soc., vol. 165, no. 3, pp. F173–F180, 2018.

Figure 1